 Examples are chlorine, phosphorus, oxygen, molten sulfur, arsenic and sodium metal. Many elements, both diatomic and regular, are hazardous materials in their elemental state. Therefore, a single atom is the smallest particle of an element that would normally be encountered. To create a smaller part of an element would require that the atom be split in a nuclear reaction. A single atom cannot be altered chemically. You cannot have a smaller piece of an element than an atom. The atom is like that last piece of paper.Ĭhlorine in its elemental form is toxic, an inhalation hazard and a strong oxidizer. Eventually, it will not be able to be cut any smaller. Then take a knife and cut the paper into smaller pieces. Keep tearing the paper in half until it becomes so small that it cannot be torn any smaller by hand. The word atom comes from the Greek, meaning "not cut." For example, take a sheet of paper and tear the paper in half. You may get chewed out afterwards by your officer or take some ribbing from other responders but no one has ever died from embarrassment! On the other hand, if you are not cautious and your errors are not on the side of safety, your actions could be fatal.Īn atom is the smallest particle of an element that can be found that retains all of its elemental characteristics. If you are going to make errors when dealing with hazardous materials, always make sure you err on the side of safety. Even if you didn't know that sodium chloride is table salt and treated it as a toxic material, your error would be on the side of safety. 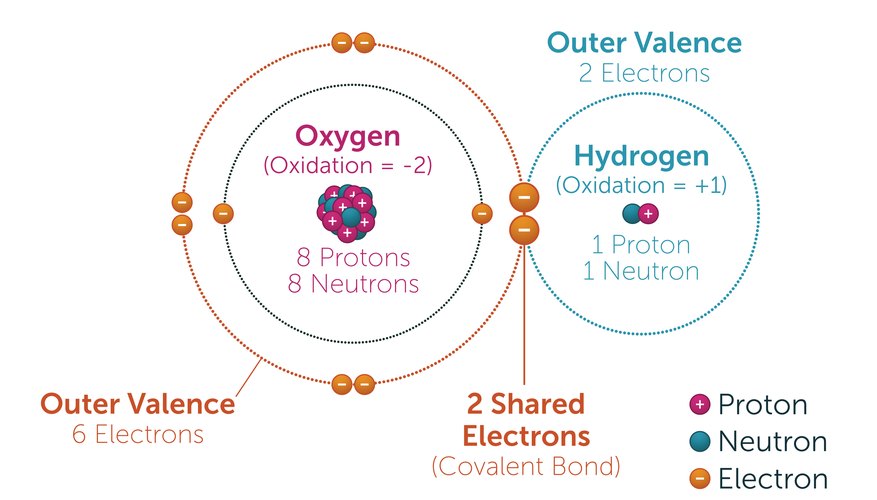
As with many rules of chemistry, there are exceptions, such as sodium chloride (NaCl) table salt. For example, generally, chemicals that contain chlorine in their formula may be toxic to some degree because chlorine is toxic. Much can be learned about a compound by looking at its elemental composition. The means of chemical bonding will be discussed later in this article. Two oxygen atoms have covalently bonded together and act as one unit. This reference to O 2 is primarily because oxygen is a diatomic element. Oxygen is commonly referred to as O 2 in emergency response. One way of remembering the diatomic elements is by using the acronym HONClBrIF, pronounced honk-le-brif, which includes the elemental symbol for each of the diatomic elements. The diatomic elements are hydrogen, oxygen, nitrogen, chlorine, bromine, iodine and fluorine. The term "di" simply means two and atomic refers to the atom. They chemically bond with another atom of that same element to form "diatomic" molecules. Some elements do not exist naturally as single atoms. The physical state of an element or compound is one of the factors that determines the appropriate level of chemical protective clothing. It is atoms of elements that combine together to form compounds of hazardous materials that responders will commonly encounter. 
The atom is the smallest part that any element can be divided into by normal means. Each symbol for an element on the Periodic Table of Elements represents one atom of that particular element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
